
Pressure Gauges
Pressure, force per unit area (N/m2), is identified as the applied force by a fluid (gas or liquid) to a surface. Pressure measurement has many applications in everyday life. For decades, tools used to measure and display pressure are known as Pressure Gauges or Vacuum Gauges. A vacuum gauge can measure the pressure ranges lower than the ambient pressure. Generally, the pressure is not an absolute quantity, and most pressure gauges determine the pressure compared to the atmospheric pressure as the reference.
Pressure Regions and Pressure Types
The vacuum pressure is defined in three regions: low vacuum pressure, high vacuum pressure (HV), and ultra-high vacuum pressure (UHV). An environment that is perfectly vacuumed is under zero pressure. But in fact, it is difficult to reach an absolute vacuum, and in practice, the pressure never becomes absolute zero.
Pressure is identified as static pressure and dynamic pressure. Static pressure is generally the same in all directions, so measuring the pressure of a static fluid is independent of the direction. On the other hand, the dynamic pressure is defined for the moving fluid, and the measurement depends on the direction of the flow. The total pressure as the sum of the static and dynamic pressures can be measured by adjusting the pressure gauge in the direction of the fluid flow. While static pressure gauges are essential for determining the net load on the pipe walls, dynamic pressure is used to measure flow velocity and air.
Pressure Gauge Types
The appropriate pressure gauge must be selected depending on the type of fluid to be measured including the range, the amount of pressure, and the environmental conditions. Pressure gauges are divided into three general groups according to their mechanism of action and type of structure: Manometers, Elastic Element Pressure Gauges, and Electrical Pressure Gauges.
Manometers
Manometers that use liquid columns to determine pressure differences are divided into four categories: Barometer, Piezometer, U Tube Manometer, and Inclined Manometer. U Tube Manometer is the simplest type consists of a U-shaped tube with one end open and the other end connected to a fluid container whose pressure needs to be measured. At first, the height of the liquid column is equal on both sides. But as the valve opens and gas or liquid enters, the height of the mercury column changes. The pressure is measured according to the change in the height of the mercury column.

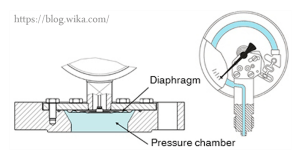
Elastic Element Pressure Gauge
Elastic element pressure gauge also includes the following four groups: Diaphragm Pressure Gauges, Capsule Pressure Gauge, Bellows Tube, and Bourdon Tube. Diaphragm Pressure Gauges can be used in the pressure range of 10 to 40 mbar to measure the pressure of gases and liquids. The measuring element consists of a circular diaphragm that is closed between a pair of flanges. The positive or negative pressure applied to this diaphragm causes deformation which is proportional to the pressure and leads to the movement of the display hand.
Electric Pressure Gauges
Electric pressure gauges depend on the type of transducer used are divided into several groups including Capacitive, Magnetic, Thermal Conductivity, Resonant, Strain Gauge, Piezoelectric, etc.
A Strain Gauge is a tool operate based on the Wheatstone bridge to measure the resistance changes. A flexible diaphragm, usually made of stainless steel, is placed between the vacuum and the atmosphere and moves due to pressure. This movement changes the electrical resistance of the strain gauge attached to the diaphragm and is sent to the display section as an electrical signal. This tool is widely used in measuring pressure, weight, torque, and in positioning sensors.


Thermal Conductivity Gauges or Thermocouple Vacuum Pressure Gauges operate based on the gas pressure that conducts the heat and decreases the temperature. Pirani Gauge is an example of thermal conductivity pressure gauges, consists of a metal wire (platinum filament) as a thermometer, exposed to environmental pressure. The wire warms up by an electric current flow through it, and the pressure of any surrounding gas can change its temperature. In general, an increase in the density of gas molecules may indicate an increase in pressure, and subsequently, the pressure increase causes thermal conduction.
With the help of a thermocouple or thermometer (metal wire), the rate of temperature reduction can be measured. The rate of temperature change depends directly on the rate at which the heat is lost according to the surrounding gas. These pressure gauges can work in the pressure range of 10-3 to 10 Torr. However, as seen, they also show sensitivity to the chemical composition of the gases. So, they mostly operate based on a relative pressure measurement method in which they can determine the pressure from the thermal conductivity of the gas molecules.
Ionization Pressure Gauges
Ionization Pressure Gauges are the most sensitive type of pressure gauge for high vacuum ranges. They indirectly measure pressure by measuring the ions produced during the electron bombardment of the gas molecules. The lower pressure, the fewer ions are produced. These pressure gauges are suitable for the 10-10 to 10-3 Torr pressure range. Ionization pressure gauges are divided into two categories: Hot Cathode and Cold Cathode.
In hot cathode pressure gauges, the electron beam is produced by heated filaments, while in cold cathode pressure gauges; the electron beam is produced by high voltage discharge. Systems manufactured based on cold cathode ionization pressure gauges have a longer life than hot cathode and operate in a pressure range of less than 10-6 Torr. They consist of a pressure gauge made by Leybold that combines the cold cathode ionization principle with the Pirani measurement system.
Pirani sensors as Micro-Electro-Mechanical System (MEMS) consist of a heated resistive element made of silicon chip used to measure the thermal conductivity. The sensor’s geometry includes the silicon chip as one surface and the cover on top of the chip as the other surface of a cavity. Due to this geometry, the sensor is insensitive to the mounting position. With the help of the diffusion mechanism, the gas molecules move toward the heated element, and there the heat loss of the gas molecules is measured.
A high voltage anode, cathode, and permanent magnet are all utilized in the cold cathode. The electrons are accelerated and ionize the gas molecules while moving toward the anode. Applying a magnetic field causes electron spiral movement while crossing the magnetic field toward the anode. This movement enhances the amount of gas ionization and an electric current will be created from the ionized molecules which is a function of the pressure.
Vacuum Pressure Gauges in Our Vacuum Coating Systems
Vac Coat Ltd. is a designer and manufacturer of high-quality and reliable vacuum coating systems that utilize Leybold PTR 90 N Penningvac vacuum gauge in DST3-T, and Agilent Varian 531 Thermocouple Vacuum Pressure Measurement Gauge in DSR1 systems.

The Triple Target Turbomolecular Pumped Sputter Coater – DST3-T is a multi vacuum coater system, combines thermal evaporator and sputter coater in one single compact desktop system. The Leybold Penningvac PTR 90N Vacuum Gauge used in this system, consists of the cold cathode ionization principle with the Pirani measurement system. It can operate in the pressure range from atmosphere to 10-9 mbar at the temperature range of 0 to + 40 ₒC.

The Desk Sputter Coater – DSR1 is a high productivity tool that delivers consistent and highly repeatable results for coating noble metals in the fully automated system. The ergonomic design and small footprint of the DSR1 allows easy use of the system. The Agilent Varian 531 used in this system is a thermocouple vacuum gauge that indicates the total pressure from 10-3 to 2.0 torr. Its sensors show high resistance to mounting orientation with rapid response to pressure changes. For more information about the systems of VacCoat Ltd., refer to the company’s website.
Some of Vac Coat Products
References
- https://www.sika.net/en/products/sensors-and-measuring-instruments/mechanical-pressure-gauges/capsule-element-pressure-gauges.html#:~:text=Capsule%20element%20pressure%20gauge,form%20a%20cylindrical%20bellows%20chamber.
- http://users.telenet.be/instrumentatie/pressure/bellows-pressure-gauge.html
- https://instrumentationtools.com/strain-gauge-working-principle/
- https://www.avnet.com/wps/portal/abacus/solutions/technologies/sensors/pressure-sensors/core-technologies/capacitive/
- https://www.lesker.com/newweb/gauges/gauges_technicalnotes_1.cfm
- John F. O’Hanlon, A User’s Guide to Vacuum Technology, pp. 92–94, John Wiley & Sons, 2005. ISBN0471467154.
- Robert M. Besançon, ed. (1990). “Vacuum Techniques”. The Encyclopedia of Physics(3rd ed.). Van Nostrand Reinhold, New York. pp. 1278–1284. ISBN 0-442-00522-9.












