Outgassing in Vacuum Deposition
Outgassing is defined as the release of a gas which has been trapped, dissolved, absorbed, or frozen in a material, a challenge in the way of high vacuum deposition. In thermodynamics, gas molecules always tend to move from a place with higher pressure to a place with lower pressure, so decreasing the pressure level in a chamber faces impediments like gas load into the chamber.
In high vacuum systems, the most important source of gas load in the vacuum process is the gases that are expelled from the surfaces of objects in the vacuum chamber called outgassing.
As to reduce the incomplete bonds of surface atoms, surfaces are active sites for the adsorption of gases and vapors that are released from the surface when placed in a vacuum. This means that all surfaces, regardless of the type of material, have outgassing.
Vacuum deposition is a key factor in preparing high quality thin films, in which the pressure of deposition chamber is decreased well below the atmospheric pressure. Therefore, mean free path of particles leaving target toward the substrate is increased, which makes thin film – a layer with several nanometers to a few micrometers thickness- formation possible.
Thin films unique properties emerge in optical, electrical, dielectric applications and so on and are highly sensitive to the presence of impurities which may deteriorate their properties. In high vacuum systems undesirable particles are reduced within the chamber, resulting in more homogenous, purer thin films; however, amount of gas load into the chamber affects pumping speed, pumping time and lowest accessible pressure [1,2].
Outgassing Sources
Four main sources for the outgassing phenomenon are:
- Vaporization: The release of matter molecules from its surface
- Desorption: The release of gas molecules that have already been adsorbed on the surface of matter. This process may occur due to heat, electrical, or optical stimulation.
- Diffusion: The release of gaseous molecules from bulk of the material. These molecules, which are dissolved in the bulk of the material during the manufacturing process or when the material has been exposed to air, reach the surface of the material when the material is in a low pressure environment (such as a vacuum chamber) and release from the surface.
- Permeation: The entry of air atoms out of the chamber into it. This phenomenon occurs when the gas atoms around the vacuum chamber enter the chamber by penetrating solid walls of the vacuum chamber. This process consists of three steps. First, the atoms are absorbed by the outer wall of the chamber. In the second step, they diffuse through the bulk material and reach the surface of the inner wall of the vacuum chamber and finally are released from the inner wall in to the vacuum chamber. These four processes are shown in Figure 1.

Outgassing Materials
Outgassing rate is the amount of gas released per unit area during per unit time, which can differ up to 9 order of magnitude for different materials. Therefore, only materials are allowed to be present in a high vacuum environment if their outgassing rate is not more than a permissible limit. Some materials that should never be used in a vacuum environment are: liquids, plastics, elastomers, adhesives, porous ceramics and effects related to living organisms such as hair, nails, skin cells, fingerprints, etc. An approximation of outgassing flow rate is given by Equation (1):
Q ̇= ∑ a1h∙A / (t⁄1h)^α Equation (1)
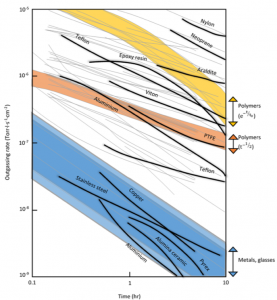
Where A is geometrical surface term, a1h is outgassing rate after 1 hour, α is decay constant, and the sum is over contributions from all the surfaces. Decay constant is in the range of 0.2-1.2 and contains information on type of material and outgassing mechanism. Decay constant of several types of materials is shown in table 1. Outgassing rate of different materials is shown in Figure 2.
| Material | Decay constant (α) |
| Clean metal surfaces | 1.1-1.2 |
| Desorption from surfaces | 1 |
| Metals, glasses, and ceramics | 1 |
| Polymers | 0.4-0.8 |
| Highly porous surfaces | 0.5-0.7 |
| Diffusion controlled outgassing from the bulk | 0.5 |
Table 1. Decay constant of some type of materials [3].
Methods of reducing outgassing:
There are several methods to lower the amount of outgassing in a vacuum chamber which include suitable choice of material, handling and cleaning the surfaces, surface treatment and baking. These methods are generally based on two approach:
1. Temporarily stimulate molecule desorption from the surface as many as possible before use,
2. Formation of a barrier on the surface to block adsorbed molecules desorb throughout operation.
Several methods to reduce outgassing are presented in Table 2.
| Method | Material | Effective for | Not Effective for | Vacuum Level | Time |
| Wash | Hot water/solvent, detergent | Gross/fine contaminants | Grease | Pressures >10-3mbar | < 30 min |
| Vapor Degrease | Heated solvent | Gross/fine contaminants, loosely bound heavy molecules | Inaccessible areas, large parts | Order of magnitude reduction in outgassing | < 1 hour |
Blowing | Air, nitrogen or other dry inert gas | Gross/fine contaminants, loosely bound heavy molecules | Grease | < 30 min | |
Chemical Treatment | H2O, HCl, HNO3or HF | Rough surfaces (O2, C, P or Cladsorption) | Order of magnitude reduction in outgassing rate | < 30 min | |
Reactive Gas | O2 or NO for oxidation, H2 or NH3 for reduction | Hydrocarbons, surface reconditioning after atmospheric exposure | Can form unwanted oxides | Reduction in outgassing rate by 50% up to 5 orders of magnitude | 30 min- 2 hours |
Glow Discharge | Ar/5-10%O2, Ar, O2, N2, H2,… | C or O based molecules, large surface areas, reconditioning after atmospheric exposure | Large components | 13 x reduction in outgassing rate | ~ 2 hours |
Passive Coating | Si, TiN, BN, Al2O3,ZrO3 | H2 from metals, CO, CO2, H2O | 0.1-100 x reduction in H2outgassing | Depends on size | |
Active Coating | Hf, Zr, Ti, Pd, V and combination | H2, H2O, CO, O2, N2, small spaces | Hydrocarbons, inert gas, continual operation (needs re-activation and replacement) | Outgassing reduced to <10−13 with no bake | Depends on size |
Vacuum Bake | Metals, UHV, H2O | Temperature sensitive components | Reduces outgassing of H2to<10−14 | 2-400 hours | |
Air Bake | Air | Stainless steel, aluminum, H2, CO, CO2, CH4 | Plastic, cadmium plating, temperature sensitive components | Reduces outgassing of H2to<10−14 | 2-400 hours |
Table 2. Methods of reducing outgassing [3].
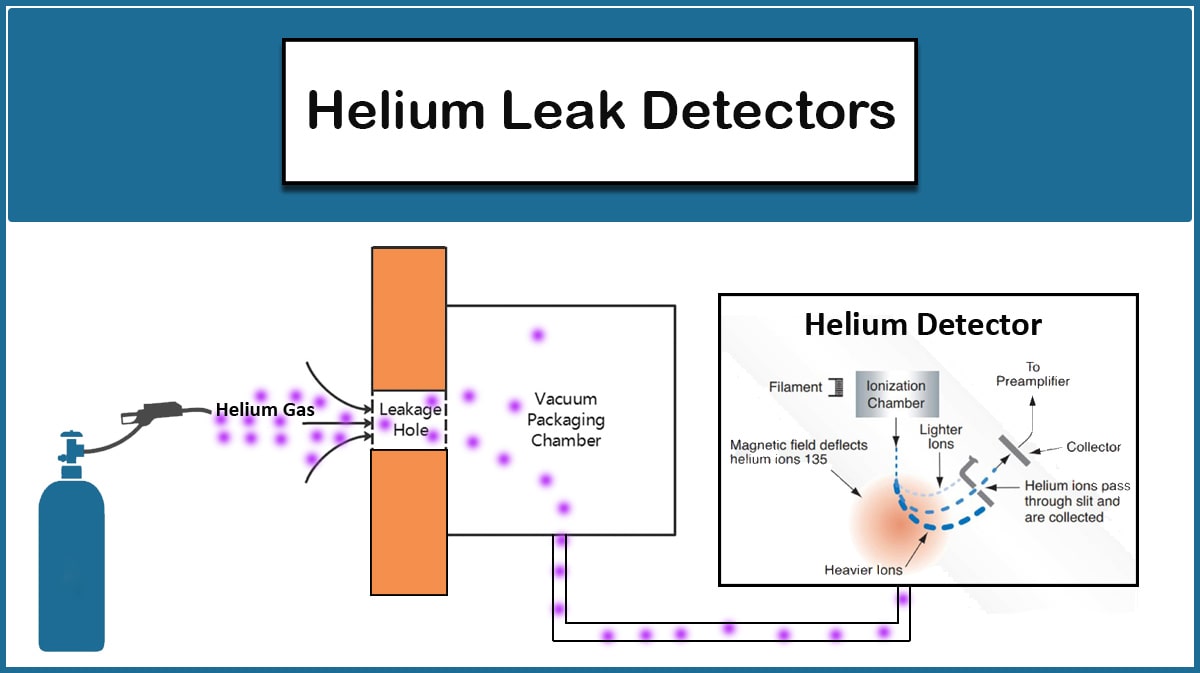
Vacuum Outgassing Vs. Leakage
The users of high vacuum systems may confuse outgassing phenomena with real leaks as the source of gas released to the vacuum chamber that obstructs the evacuating process. In the case of Outgassing, the pressure decreases over time, but in the leakage case, the pressure does not fall below a certain level.
The lower the pressure, the more outgassing occurs. This phenomenon is usually not noticeable at the pressures reached by the rotary pump. Thus discovering the source of problem that prevents the system to be evacuated is the first step to find the proper solution. Vacuum leakage source detection methods and the appropriate solutions are discussed in another article.
Many devices that operate in the very high vacuum range (UHV) use so-called electric heating blankets. In these systems, by heating or so-called baking the system, the heat energy is transferred to the molecules trapped on the inner surfaces of the vacuum chamber and they are separated from the surface faster and transferred to the outside of the vacuum chamber.
As mentioned, the material used in the construction of vacuum systems has a significant effect on reducing the outgassing phenomenon. This point has not been ignored in the production of Vac Coat products. The vacuum chamber and its components in the products of this company, which are all vacuum coating systems, are designed and built in such a way as to minimize the amount of outgassing in these systems.
Vac Coat Sputter Coaters
Vac Coat Ltd. is a vacuum coating system designer and manufacturer known for high-quality and reliable vacuum coating systems. Its products include sputtering systems, thermal evaporation systems, carbon coating systems, and pulsed laser deposition system used to create thin films with a thickness in the range of nanometer to a few micrometers. All Vac Coat Sputter Coaters deposit thin films by magnetron sputtering method.
The Sputter Coaters include triple-cathode sputter coater with a thermal evaporator (DST3 & DST3-T), magnetron desk sputter coater (DST1-300 & DST1-170), and turbo pumped desk sputter and carbon coater provided with a turbomolecular pump (DSCT) and rotary pumped desk sputter and carbon coater (DSCR), which are most favorable for SEM sample preparation. For more information, visit the website please.
References
Read the following article for more on outgassing topic:
- [1] A. Jilani, M. S. Abdel-wahab, and A. HosnyHammad, “Advance Deposition Techniques for Thin Film and Coating”, in Modern Technologies for Creating the Thin-film Systems and Coatings. London, United Kingdom: IntechOpen, 2017 [Online]. Available: https://www.intechopen.com/chapters/52684 doi: 10.5772/65702
- [2] A. Roth, Vacuum Technology, Third Edition, Elsevier, North Holland (2012)
- [3] A Review of Outgassing and Methods for its Reduction, Appl. Sci. Converg. Technol. 26(5): 95-109 (2017) http://dx.doi.org/10.5757/ASCT.2017.26.5.95
- Outgassing rate of different materials and its measurement methods, International Journal of Scientific & Engineering Research, Volume 8, Issue 4, April-2017, ISSN 2229-5518
- https://www.shinmaywa.co.jp/vac/english/vacuum/vacuum_2.html