
What is the Purpose of Sample Preparation?
Samples to be investigated by electron microscopy need preparation through various techniques to obtain the most accurate results. The sample preparation procedure may include steps such as washing, drying, fixation, mounting, and coating to achieve the best result. Not carrying out preparation steps before imaging may deteriorate the device’s performance, image quality, and sample structure.
What is SEM?
Scanning Electron Microscopy (SEM) is a powerful tool to achieve information on the structure and other nanoscale characteristics of a specimen. This type of electron microscopy provides nanoscale images which can be used to study various samples, including ceramics, metals, alloys, semiconductors, biological samples, etc.
Why Should an SEM Sample be Prepared?
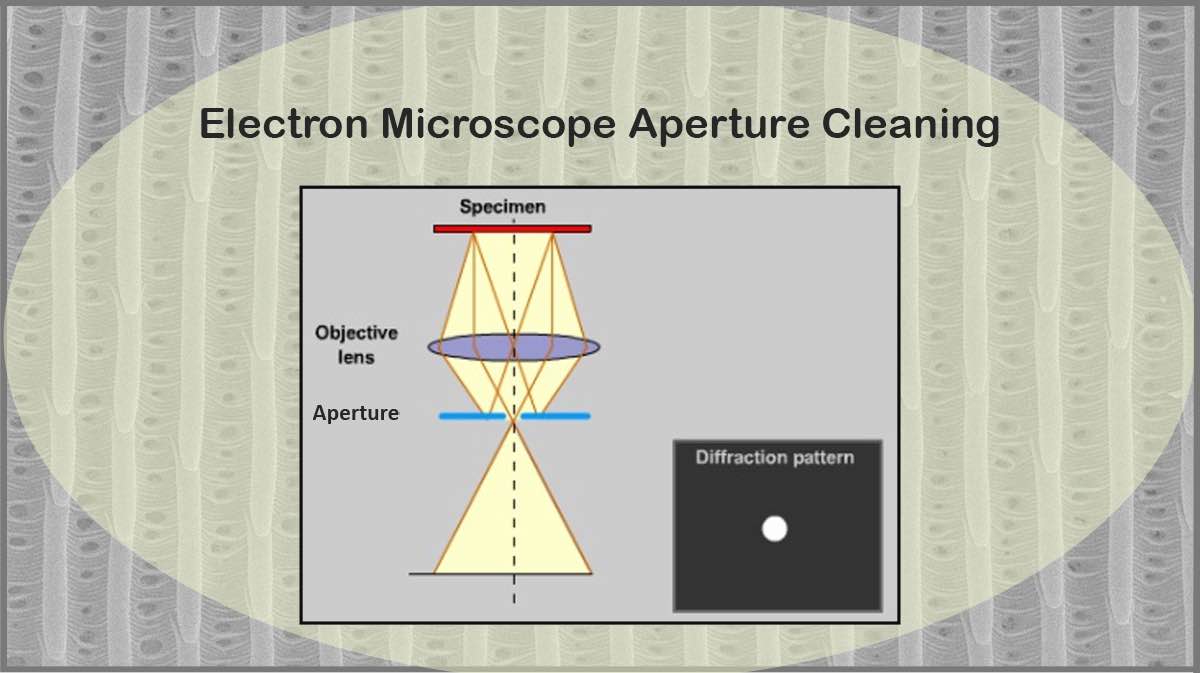
SEM samples should be dry and conductive. Samples containing water cause problems when vacuuming the SEM chamber, also energetic electrons incident on the sample may heat it, which can result in outgassing and increasing chamber contaminations, leading to deterioration of image quality. Also, heating up the sample by the electron beam can damage the sample surface. In case of imaging insulating samples, the conductivity of the sample surface should be improved by a very thin conductive coating to prevent charge build-up on it (Figure 1).

Coating the electron microscope samples have other advantages like reducing the volume of penetration, which guarantees the resulting image merely displayes the topmost surface structure of the sample, and electrons are not from the deeper layers. Consequently, the secondary electron (SE) emission and the signal to noise ratio could be enhanced in the coated samples, resulting in high-resolution, high-contrast imaging.
Accordingly, some specimens require a preparation step before SEM imaging.
SEM Sample Preparation Process
The SEM sample preparation mainly depends on the sample characteristics, it may need some or all of the following steps (Figure 2):
- Cleaning and cutting
- Fixation and stabilization
- Dehydration
- Drying
- Mounting on a stub
- Coating with a conductive material
SEM Sample Preparation Techniques
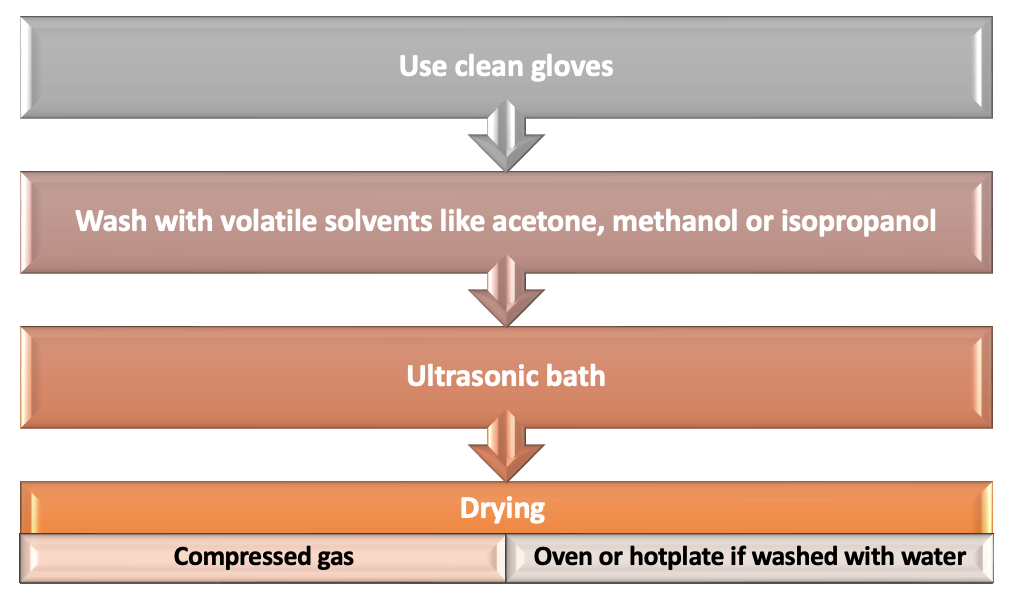
Sample Cleaning and Drying
Cleaning the samples is a crucial step in the sample prepapration process to obtain clear images. Degreasing and drying the sample reduces outgassing from possible organic contaminations and residual water in the sample. Some samples like polymers, minerals, clays, zeolites, and textiles, that may contain moisture or trapped gas, need to be outgassed before EM imaging. To clean and dry a sample one can follows the below steps:
- Wash the sample with volatile solvents like acetone, methanol, or isopropanol
- Use ultrasonic bath (High power baths are not recommended to prevent any physical damage to the sample)
- Drying using compressed gas
- If cleaned by water, dry the samples using hot plate or oven
- Use clean gloves during sample preparation and handling (Hand grease is a major outgassing source in a vacuum system)
Some samples should be cut, polished, or etched (Like plasma etching) prior to be mounted onto the SEM stub.
Also, the vacuuming process should be considered carefully for delicate, outgassing samples, since they may endure fractures and shape shifting under quick pumping down procedures, resulting in change in their appearance.
Biological Samples
Biological samples should undergoe fixation and dehydration processes, such as Critical Point Drying (CPD) or freezdrying, and then dried to remove the water inside them. This will allow higher vacuum SEM imaging and clearer images. Moist samples that can not be dehydrated can be imaged by cryo-SEM systems.
Powder Samples
Powder samples may fly off under vacuum condition inside the SEM chamber, so they must be processed. Large amount of powder can be compressed to a pill. Small amount of micron-sized powders can be spread onto a carbon tape, followed by N2 gas flow to remove extra powders. Nano-sized powders should be completely dispersed in a volatile solvent, by ultra-sonication or other methods, then a small droplet of them is dropped on a suitable substrate to dry. Vacuum filteration is another way to prepare a powder for SEM imaging.
Non-Conductive Samples
Non-conducative samples (including biological ones) can trap incident beam electrons on the surface and result in shinning spots in SEM images. Also this built-up charge can cause overheat and deterioration of the sample. Coating a thin film of a conducting material on the sample surface can prevent this phenomena (Figure 3).
The coating thickness must be enough to prevent charging (You can read more about thickness uniformity here), while preserving specimen surface details (around 10 nm). Deposition of a layer of gold, silver, platinum, or chromium for SEM and carbon for EDX are ideal, since carbon spectral lines in EDX normally has less interference with the sample’s compared with gold. For more details on the coating material characteristics visit our blog article Selection of suitable material for preparing an electron microscope sample.
Coating Techniques on SEM Specimens
Sputtering Deposition
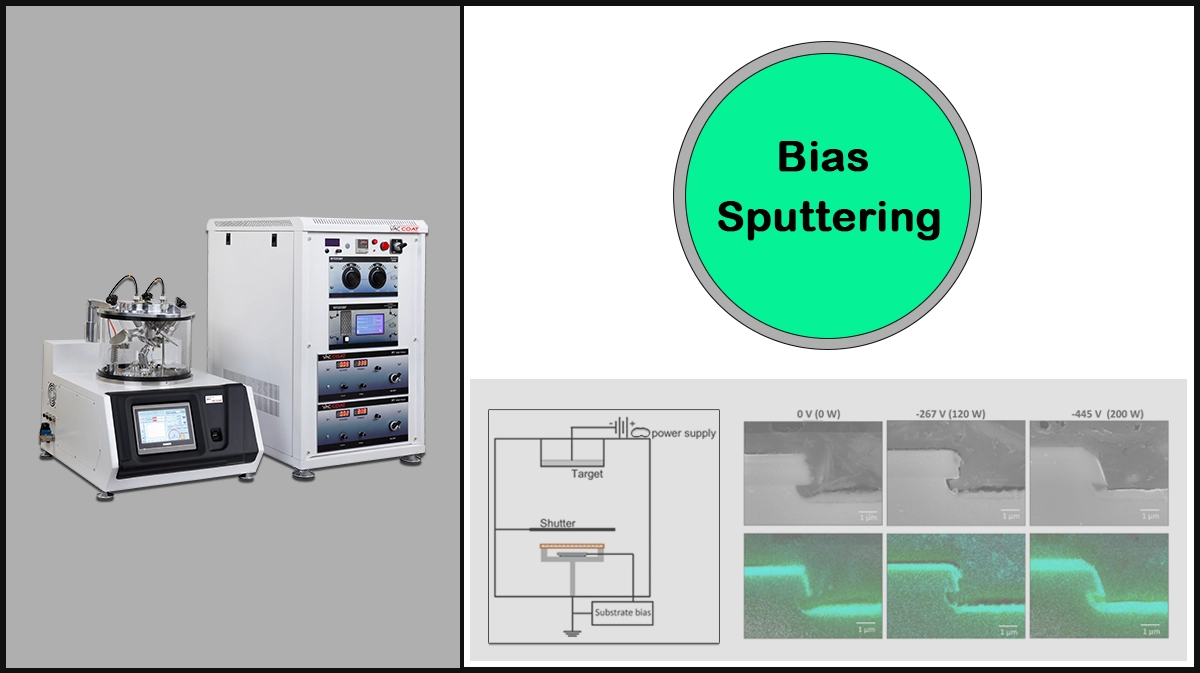
The traditional Diod Sputtering uses high-sputter yield metals, i.e., nickel, gold, gold-palladium, and platinum, has the disadvantage of high secondary energy output through released electrons, which can be overcome by applying Bias Sputtering. Other sputtering techniques, such as Magnetron Sputtering and Ion Beam Sputtering have been developed to enable deposition of metals like Pt, W, Ta, and Cr.
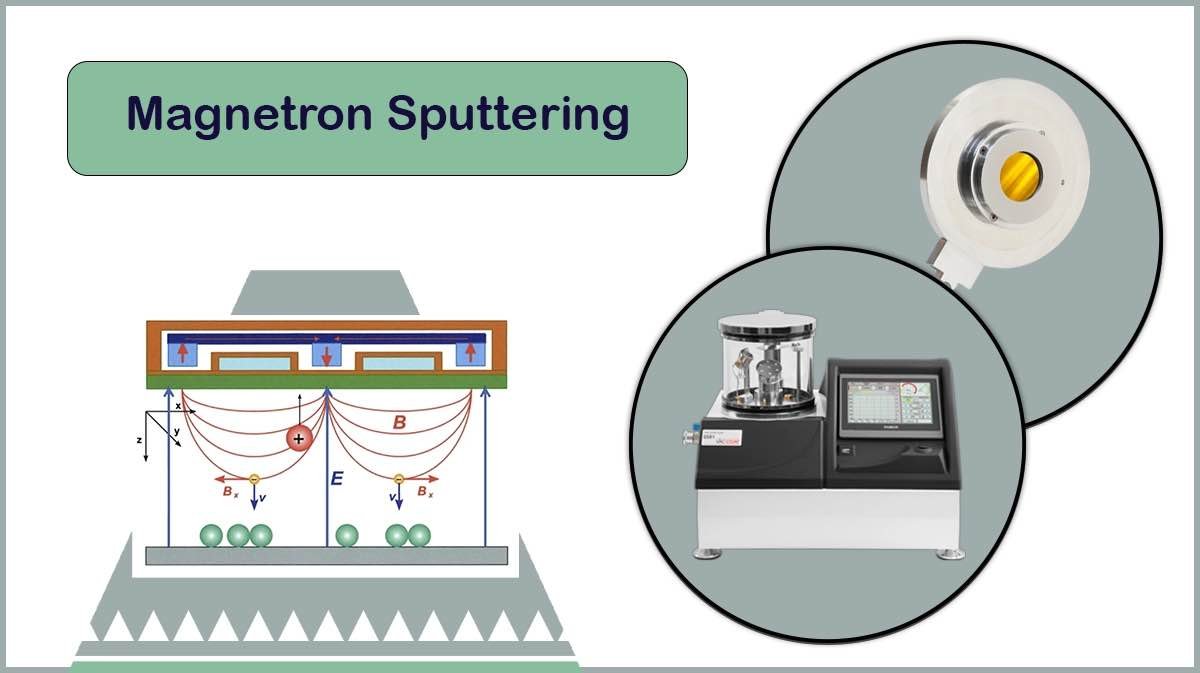
– Magnetron Sputtering
In magnetron sputtering, a sputter cathode with magnets is utilised to apply a magnetic field over the target, confining the plasma particles. This technique allows deposition of fine-grained coatings of metals like Chromium or Tungsten, in a high-vacuum, low-temperature environment, resulting in high-resolution, conductive layers ideal for FE-SEM imaging, as provided by Vac Coat high-vacuum sputter coaters, DST1-170 and DST1-300.
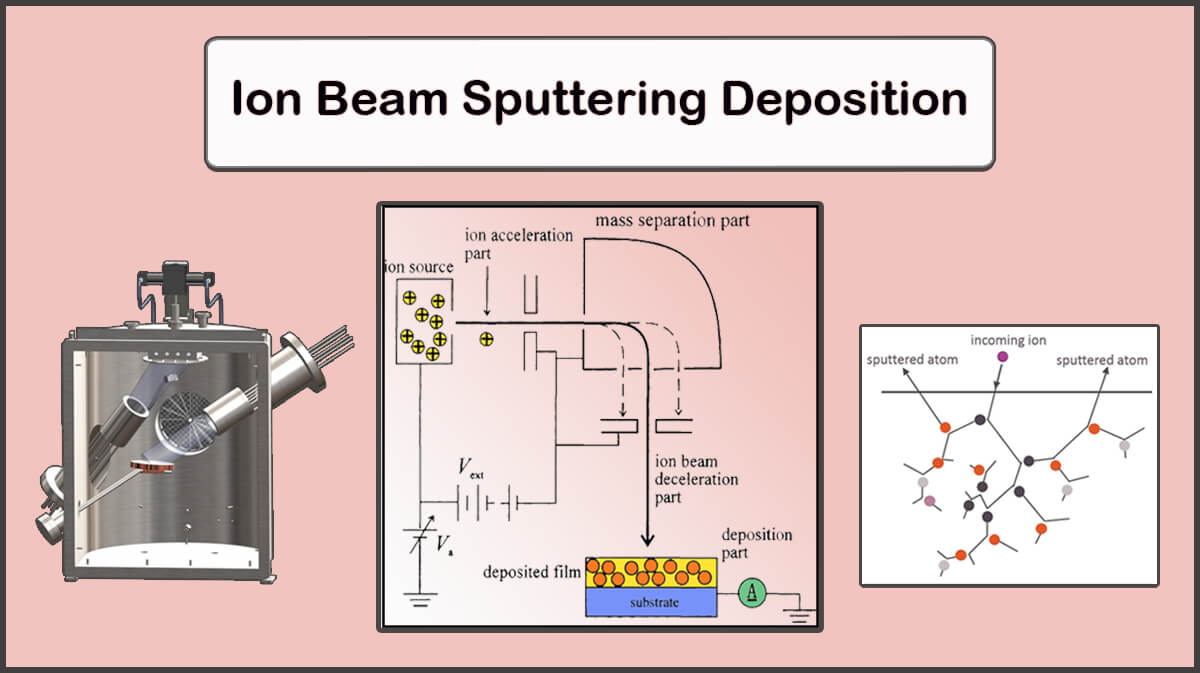
– Ion Beam Sputtering Deposition (IBSD)
The ion beam sputtering can provide fine-grain structures with extremely low thicknesses (Below 1 nm) in a low temperature chamber to improve imaging results of the electron microscopy of bio-specimens.
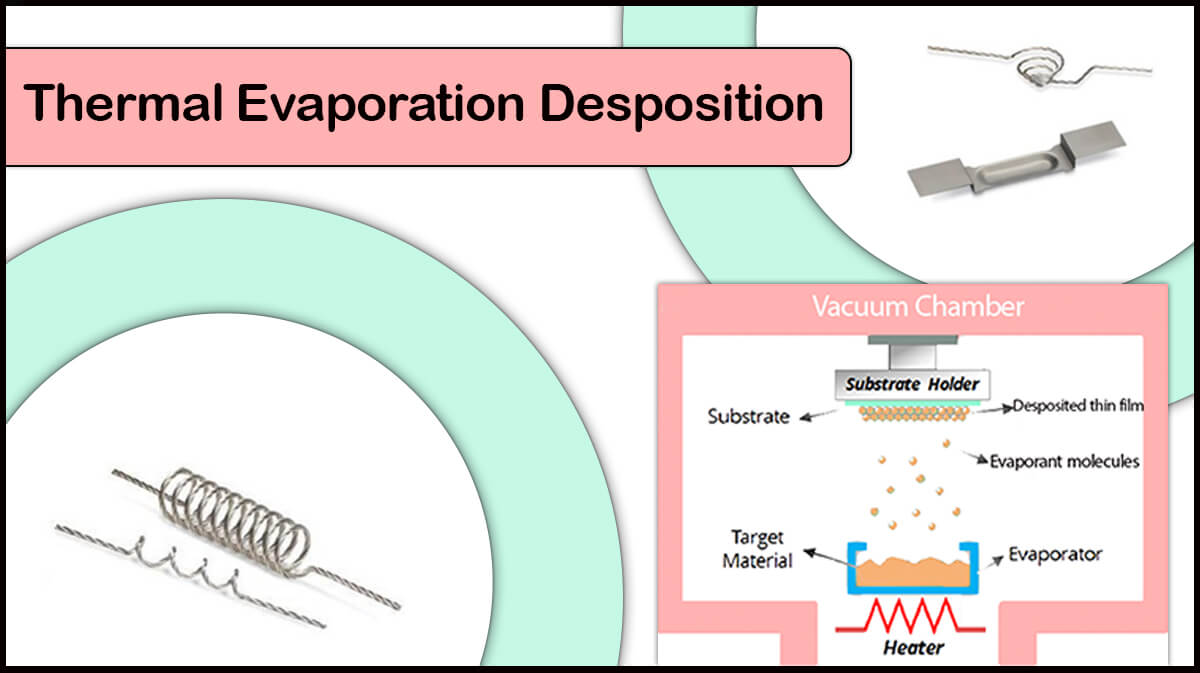
Evaporation
Thermal evaporation and E-beam evaporation of metal or carbon sources are applicable for SEM coating to provide fine-grain structure coatings on the specimen.
Carbon coating by thermal evaporation (thread or rod) is frequently used for SEM/FE-SEM imaging, accompanied by EDX/EDS analysis, as the X-ray spectrum of carbon does not interfere with the sample, due to its low atomic number. Vac Coat carbon coaters, including DCR, DCT, and DCT-300, as well as hybrid coaters like DSCR and DSCT, enable thin film carbon coatings for SEM, FE-SEM, and EDX analysis, where the high vacuum systems (DCT, DSCT, and DCT-300) empower coating of more amorphous layers for high-resolution analysis.
Metal Shadowing (Shadow-Casting at Oblique Angle)
The shadow casting of a biological sample is typically performed by evaporation or sputtering deposition of a high-density metal at an oblique angle onto the specimen, resulting in a uniform, structureless, extremely thin film (<0.1 nm thick).
How does an oblique angle help to image fine structures?
Smaller objects should be shadowed at smaller angles. Deposition at oblique angles coats the relatively higher portions of the specimen, which casts a partial or total shadow in the direction opposite the source.
SEM/TEM imaging of shadow-casted surfaces
The metal shadowed specimens are observable through the electron microscope, SEM or TEM. The thicker metal-coated areas will scatter more of the electron beam and appear as darker regions in TEM, whereas the thinner coatings will be transparent to the beam and appear bright in TEM. For SEM, it is quite the opposite.
Shadow-casting methods: Unidirectional and Rotational
Unidirectional shadow-casting is applicable to investigate soft biological surfaces and wetting behavior of micro- and nanoparticles at liquid-liquid interfaces by the Freeze-fracture Shadow-Casting (FreSCA) method.
Using rotary shadowing techniques, in which the specimen is placed on a rotary sample holder, results in 3D visualisation and surface detail of viruses, bacteria, proteins, or DNA.
Figure 5. Shadow casting thin films of Platinum/Carbon layers on a surface with an intricate structure
To conclude, the choice of the best preparation method for samples to be imaged by SEM strongly depends on the type of microscopy, imaging conditions, and the required imaging resolution.
Vac Coat Products & SEM Sample Preparation
Vac Coat produces high-precision coating systems for a variety of purposes. Vac Coat SEM coaters can creat thin condictive films on SEM samples for enhanced SEM/FESEM imaging. Vac Coat sputter coaters equipped with vacuum rotary and turbomolecular pumps, working at low and high vacuum conditions (DSR1 and DST1), are a suitable choice for SEM sample preparation.
Vac Coat also provides low and high vacuum carbon coaters (DCR and DCT) for carbon deposition. Hybrid sputter coater and carbon coater (DSCR and DSCT) systems offer switching between these two techniques in one desktop deposition system. DTE and DTT thermal evaporators can also be used for coating on SEM sample.

High-precision QCM thickness meter, electronic shutters, sample rotation, 7” colored touch-screen panel, user-friendly updatable software, and many other special features are presented in these benchtop coating systems. DST1 is presented in two chamber size, with a diameter of 170 or 300 mm. Optional features, such as RF sputtering, plasma cleaning, and substrate bias voltage and heating can be offered due to customer’s requirements.
Some of Vac Coat Products
References
- Polilov, A.A., Makarova, A.A., Pang, S. et al. Protocol for preparation of heterogeneous biological samples for 3D electron microscopy: a case study for insects. Sci Rep 11, 4717 (2021). https://doi.org/10.1038/s41598-021-83936-0
- Echlin, Patrick. Handbook of sample preparation for scanning electron microscopy and X-ray microanalysis. Springer Science & Business Media, 2011.
- Hendricks GM. Metal shadowing for electron microscopy. Methods Mol Biol. 2014;1117:73-93. doi: 10.1007/978-1-62703-776-1_5.
- Peters, K-R. “Metal deposition by high-energy sputtering for high magnification electron microscopy.” Advanced Techniques in Biological Electron Microscopy III. Berlin, Heidelberg: Springer Berlin Heidelberg, 1986. 101-166.
- Isa, L. (2013). Freeze-fracture Shadow-casting (FreSCa) Cryo-SEM as a Tool to Investigate the Wetting of Micro- and Nanoparticles at Liquid–Liquid Interfaces. CHIMIA, 67(4), 231. https://doi.org/10.2533/chimia.2013.231.
- http://www9.open.ac.uk/emsuite/services/sem-and-tem-sample-preparation-materials
- https://ywcmatsci.yale.edu/facilities/sem/sample-preparation
- Murtey, Mogana, Ramasamy, Patchamuthu. “Sample Preparations for Scanning Electron Microscopy – Life Sciences”. Modern Electron Microscopy in Physical and Life Sciences, edited by Milos Janecek, Robert Kral, IntechOpen, 2016. 10.5772/61720.
- https://www.gu.se/en/core-facilities/sem-sample-preparation-techniques
- Nguyen, Jenny Ngoc Tran, and Amanda M. Harbison. “Scanning electron microscopy sample preparation and imaging.” Molecular Profiling. Humana Press, New York, NY, 2017. 71-84.
- https://bitesizebio.com/34150/sem-sample-prep/#fl-main-content





















After completion of fixation and dehydration steps for preparing a sample for SEM analysis, is it possible to store it in drying stage for a while, before mounting on a stub?
To store a dry sample for a long time and preserve its structural features, it should be stored in a low humidity environment like a dessicator.
Hello all, How to prepare a powder of a non-conducting material, like liposom, for scanning electron microscopy?
Hello. The main method is to spread a small amount of powder on one side of a conductive dual-sided adhesive tape, such as carbon or copper tape. Also, sputter coating of a thin gold layer (a few angstroms) on the powders results in a better electrical conductivity and clearer SEM images.
How to prepare greases like lithium-calcium complex grease for SEM imaging?
“SEM analysis requires a high vacuum in the sample chamber, so it is necessary to extract the oil from the lubricants and grease to image their morphology in SEM characterization. The conventional sample preparation steps include:
1- Oil extraction by immersing the sample for one week in an organic solvent.
2- Replacing the solvent 3 – 4 times per day until oil extraction is completed.
3- Drying the samples at room temperature for several days.
4- Coating the samples with a thin layer of gold by means of a sputter coater to prevent charging of the surface, reduce thermal damage, and improve the secondary electron signal required for topographic examination in the SEM.”
Hi sir, how to draw a filtration sample onto filter paper and mount it on the sem stub
The methods may vary due to the type of sample characteristics, like size of the nanoparticles, solubility, etc., according to the scientific reports.
Hi, how do I prepare Biochar sample for SEM
Biochar is a carbon (C) rich material produced by thermal decomposition of biomass under oxygen (O2) limited conditions. Due to its carbon rich structure, it seems it should be conductive, and no further conductive coating for SEM sample preparation is needed. However, more detailed information is available in the scientific literature.
How do I prepare Biochar sample for SEM?
Biochar is a carbon (C) rich material produced by thermal decomposition of biomass under oxygen (O2) limited conditions. Due to its carbon rich structure, it seems it should be conductive, and no further conductive coating for SEM sample preparation is needed. However, more detailed information is available in the scientific literature.
can i use the same method for preparing sample of agrowaste?
The agro waste is kind of biological samples that should be treated and prepared in the same way, including steps like fixation, dehydrstion, thin film of metal coating, etc.
The agro waste is kind of biological samples that should be treated and prepared in the same way, including steps like fixation, dehydrstion, thin film of metal coating, etc.
Can we achieve even clearer SEM images by manipulating the conductivity of the sample through unconventional means, such as incorporating unique materials or altering the composition during the preparation process?”,
“refusal
Basically, the SEM imaging aims to reveal the surface structure of the specimen. The SEM coaters deposit a very thin film of conductive materials, using sputtering deposition of metals or carbon coating, to maintain the original surface structure, as well as increasing its electrical conductivity. So, manipulating samples’ surface composition and structure to make them more conductive may undesirably deteriorate the original surface properties under-study.
Elis Thal
Hi Sir,
Can the DST1-300 apply a multilayer metallization composed of such as Ti/Pt/Au on surfaces like ceramic or glass in a single run?
Hi, DST1-300 is a single cathode sputter coater. If subsequent deposition of materials (up to 3 targets) in a single run is required, the Vac Coat triple-cathode DST3 model will suit best.
Hi,
Do you have any advice on how to attach small delicate, biological samples such as infusoria (e.g. paramecium), cyanophytes filtered from water samples to glass or mica slips.
I have tried using both silanised and chromium gelatine slides but have only ever managed very poor and unreliable results. Any help would be greatly appreciated.
Vac Coat Ltd. offers vacuum coating systems for thin film deposition purposes. Powder samples can be deposited onto a substrate by the thermal evaporation process. However, deposition of biological sources through the evaporation process may damage the material structure and is typically not recommended. Some of the steps mentioned in our blog article, sample preparation for the electron microscopy analysis, may help to achieve the goal.
what is name of method which we used for prepration the samples?
Carbon coating by high-vacuum carbon evaporation for sample preparation.